Chemistry
Pneumatic Trough, Glass, 200 x 100 mm
Circular pneumatic trough, made from clear soda-lime glass.
194.86 AED
Pneumatic Trough, Glass, 250 x 130 mm
Circular pneumatic trough, made from clear soda-lime glass.
259.28 AED
Pneumatic Trough, Glass, 300 x 130 mm
Circular pneumatic trough, made from clear soda-lime glass.
280.63 AED
Gas Washing Bottle, Dreschel
The head is fitted with 8 mm diameter inlet and outlet tubes.
142.29 AED
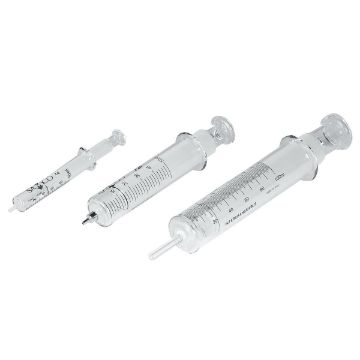
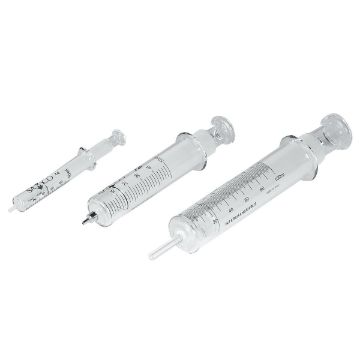
Gas Syringe, 100 mL
Ground glass syringe. Marked with 20 mL major graduations, and 1 mL minor graduations. Suba-Seal Stoppers ST14710 or ST14715 can be used as resealable stoppers. For video demonstration, visit timstar.co/SubaSealSyringe
223.71 AED
Delivery Tube, Polypropylene, 90°
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Eight types available. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. ** please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
183.70 AED
Delivery Tube, Polypropylene, Bung Top
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Eight types available. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. ** please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
183.70 AED
Delivery Tube, Polypropylene, Bung Top 2
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Eight types available. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. ** please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
193.00 AED
Delivery Tubes, Polypropylene, Straight
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
164.30 AED
Delivery Tubes, Polypropylene, Small U-Bend
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
193.00 AED
Delivery Tubes, Polypropylene, Medium U-Bend
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
183.70 AED
Delivery Tubes, Polypropylene, Large U-Bend
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
211.60 AED
Delivery Tubes, Polypropylene, Side Arm U-Bend
Pre-bent rigid polypropylene delivery tubes. Unbreakable in normal use ideal substitutes for fragile glass alternatives. Suitable for many types of gas collection experiments. Supplied in pack of 5. The tubes have an i.d. of 4 mm, and an o.d. of 6 mm. please note these are not suitable for use at higher temperatures as this will distort the polypropylene.
202.30 AED
Universal Electrode Holder
These versatile electrode holders are suitable for holding Carbon rod electrodes as well as metal strip electrodes. They are designed to fit over 100 mL and 250 mL glass beakers, being held in place by small plastic lugs to prevent them slipping off. Resistant to corrosion, they accept 6 - 8 mm diameter cylindrical (e.g. carbon) electrodes and 19mm wide, 0.8 mm (minimum) thick metal strip electrodes. The brass connectors also resist corrosion and are compatible with the usual 4 mm stackable type lab leads. To tighten the carbon electrodes without crushing them, the setscrew is equipped with stop. • 4 mm sockets • Dimensions (L x W x H): 100 x 40 x 15 mm • Complete with 2 carbon electrodes
166.01 AED
S Shape Electrodes
These 'S' shaped electrodes are convenient and easy to use simply hang them over the rim of the beaker containing the electrolyte and connect crocodile clips with 4 mm leads to them! Made from insulated brass, with a short length of carbon rod attached at the lower end and bare contact at the top for attaching crocodile clip. Supplied as a pair of electrodes.
54.54 AED
Electrodes, Round, Copper, 150 mm
These round electrodes will fit the electrode holder EL110620, the electrode connector EL150426, and rubber stoppers with hole. Diameter 6-7 mm.
37.15 AED
Electrodes, Platinum, with Screw Terminals
Platinum electrodes, with screw terminals. Manufactured in borosilicate glass. Length from tip of foil to tip of terminal, 114 mm.
679.83 AED
Electrodes Round, Aluminium, 150 mm
These round electrodes will fit the electrode holder EL110620, the electrode connector EL150426, and rubber stoppers with hole. Diameter 6-7 mm.
29.25 AED
Electrodes Round, Nickel, 150 mm
These round electrodes will fit the electrode holder EL110620, the electrode connector EL150426, and rubber stoppers with hole. Diameter 6-7 mm.
86.56 AED
Electrodes Round, Carbon, 200 mm
These round electrodes will fit the electrode holder EL110620, the electrode connector EL150426, and rubber stoppers with hole. Diameter 6-7 mm.
209.48 AED
Electrodes Round, Iron, 150 mm
These round electrodes will fit the electrode holder EL110620, the electrode connector EL150426, and rubber stoppers with hole. Diameter 6-7 mm.
33.20 AED
Electrodes Round, Tin, 150 mm
These round electrodes will fit the electrode holder EL110620, the electrode connector EL150426, and rubber stoppers with hole. Diameter 6-7 mm.
76.68 AED
Electrodes Round, Zinc, 150 mm
These round electrodes will fit the electrode holder EL110620, the electrode connector EL150426, and rubber stoppers with hole. Diameter 6-7 mm.
49.80 AED
Electrolysis Cells
Each cell consists of 75 x 25 mm glass cylinder with 2 hole rubber stopper fitted with carbon electrodes. Electrical contact is made through the crocodile clips and a piece of insulating material between the electrodes prevents short circuiting. The gases evolved are collected in two 75 x 10 mm test tubes suspended over the electrodes by piece of wood and rubber bands (not supplied).
218.18 AED
Fruit Clock
This kit uses the juice within lemon as an electrolyte in an oxidation-reduction reaction to produce useful electricity sufficient to power clock. The electrodes must be sanded clean between each attempt. Requires lemons to make ""batteries"" in series to generate sufficient voltage to power the clock.
74.31 AED
Salt Bridge
Consisting of an inverted U-tube with its ends closed by sintered discs. Dimensions (Diameter W x H): Arms 13 x 80 x 100 mm.
112.65 AED